Scientific Achievement
An international team of scientists working at the Molecular Foundry has revealed how interactions between electrons and ions can slow down the performance of electrodes made with the compound vanadium pentoxide.
Significance and Impact
Vanadium pentoxide’s layered atomic structure results in a vast surface area that may lead to batteries that pack more punch, last longer and charge or discharge more quickly.
Research Details
- Vanadium pentoxide’s potential has been limited by its subpar performance when put to the test in the real world.
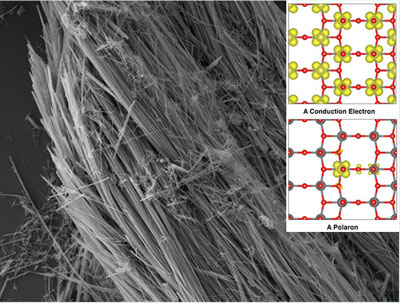
- Molecular Foundry users made 2D maps of the electronic properties of synthesized vanadium pentoxide nanowires serving as a model lithium-ion cathode using Scanning Transmission X-ray Microscopy at The Canadian Light Source. They came to the Molecular Foundry to interpret their findings.
- These nanoscale measurements provided evidence that electrons in vanadium pentoxide sit predominantly on vanadium atoms near lithium ions and induce a distortion in the surrounding crystal structure.
- The combination of a charged particle and an associated local structural distortion is referred to as a “polaron.”
- The polaron, or the localization of electron density, slows down the
movement of electrical current and ions.

