Scientific Achievement
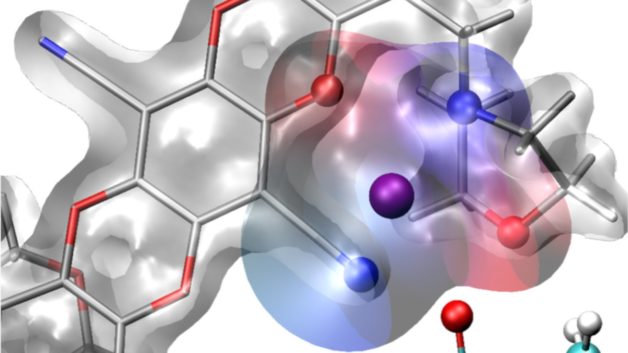
Foundry scientists and users designed a new polymer membrane containing molecular cages that can selectively bind lithium ions and also enhance their diffusion through the membrane.
Significance and Impact
The team used a workflow from drug discovery to identify the new polymer, which increased the flow of ions by an order of magnitude. The new membrane could enable more powerful and efficient batteries, and the design method opens the door to discovering polymers for other clean energy applications.
Research Details
- The research team was inspired by a widely-used drug discovery process and built and screened large libraries of membranes with diverse pore structures in order to identify those capable of binding to lithium ions.
- They identified a polymer containing solvation cages built into its pores that selectively bind lithium ions and enable their rapid transport through the membrane.
- Simulations performed at NERSC revealed how the lithium ions bind to the solvation cages and X-ray studies at the ALS showed that the new membrane enabled rapid and efficient charging and discharging compared to a standard membrane.

