Scientific Achievement
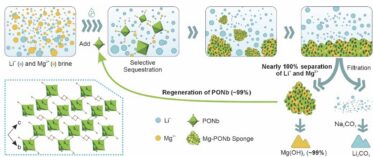
Researchers developed a new material that selectively captures magnesium ions from a Lithium-rich brine, achieving over 99.9% magnesium removal in under one minute.
Significance and Impact
This technology could dramatically improve lithium extraction for batteries and electric vehicles while simultaneously recovering valuable magnesium that is currently discarded as waste.
Research Details
- Polyoxometalates (POMs) offer a tunable platform for selective ion separations. The researchers used a “Mg-PONb sponge,” a [Nb6O19]8−-based system that selectively sequesters Mg2+ from Li+, Na+, and K+ in brines.
- Spectroscopic, crystallographic, and computational analyses revealed that strong Mg2+ coordination to terminal oxygen sites in the PONb cluster drives the formation of a structurally distinct porous framework, enabling >99.9% Mg2+ exclusion within seconds while achieving nearly 100% separation efficiency for both Li+ and Mg2+.
Chen, L., Li, C., Adibnia, S., Yang, S., Li, J., Samolova, E., Dopilka, A., Huang, Z., Giovine, R., Fleming, X.B., Guo, J., Haddad, A.Z., Kostecki, R., Chen, W,. Dun, C., Urban, J.J. Sci. Adv. 11, 49. (2025) DOI:10.1126/sciadv.adz7696
Research Summary
The researchers addressed a practical problem in lithium extraction from brine sources: separating lithium from magnesium, two chemically similar ions that typically occur together in natural brines. Current extraction methods are slow and inefficient, recovering less than 40% of available lithium while discarding magnesium as waste. The team developed a material based on polyoxoniobates, specifically a niobium-based cluster compound called [Nb6O19]8−, that selectively captures magnesium ions. When this material contacts brine, it rapidly forms a porous framework that binds magnesium through coordination bonds with oxygen atoms on the cluster surface, while allowing lithium ions to pass through. The process completes in under one minute and works effectively even when magnesium concentrations are up to 200 times higher than lithium concentrations, which is typical in real brines. The material removed over 99.9% of magnesium with minimal lithium loss and achieved lithium-to-magnesium selectivity ratios exceeding 5000. The material can be regenerated and reused multiple times without performance degradation, and the captured magnesium can be recovered as crystalline magnesium hydroxide. This work addresses a key limitation in meeting growing lithium demand. Current methods for extracting lithium from brine (which contains about 60% of global reserves) are inefficient. The new technology could improve lithium production rates while reducing extraction costs. The method also enables recovery of magnesium as a useful product rather than waste, which improves the economics of brine processing since magnesium is needed for automotive and aerospace applications.

