Scientific Achievement
Molecular Foundry staff and users have combined to develop membranes for lithium-sulfur batteries made from polymers of intrinsic microporosity (PIMs) that are 500 times more effective than conventional membranes.
Significance and Impact
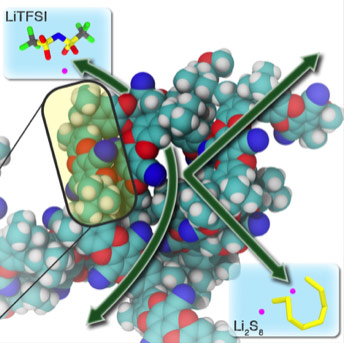
Lithium-sulfur batteries are well poised to provide high-density, long-term and low-cost electrochemical energy storage but current designs have limited efficiency and lifetimes due to the uncontrolled migration of soluble sulfur species through the membrane that separates the electrodes.
Research Details
- The ideal battery membrane achieves high-flux, ion-selective transport in non-aqueous electrolytes.
- Organic chemists, guided by theoretical calculations, developed PIMs that feature pore sizes of less than 1 nm in diameter, compared to the 17 nanometer pore size of typical membrane separators.
- Smaller ions, like lithium and sodium are allowed to pass through the membrane while larger polysulfides are blocked.
- The PIMs are synthesized in a single step and easily cast into large-area sheets.

