By Jeremy Snyder
In the quest to understand our brains, a long-sought goal for researchers has been the ability to “talk” to neurons: to tell specific brain cells to activate, and be able to listen in real time as they react. Now, a team of Foundry users and staff have unlocked that ability in an unprecedented way by manipulating light at the nanoscale.
The effort, led by Foundry user Vittorino Lanzio and Nanofabrication Facility Director Stefano Cabrini, has produced a new kind of neural probe that packs the ability to both excite and monitor neuron activity inside an exceptionally small package, an order of magnitude more condensed than the current state of the art.
Neural probes are needle-like instruments that are inserted into a brain to monitor its activity in-vivo by picking up on the electrical current from nearby neurons. They fill an important niche in the world of neuroscience by allowing researchers to look at activity deep within the brain of a living animal, in real time. The best neural probes also contain mechanisms for triggering that activity on-demand using drugs, electricity, or light. Of these stimuli, photo-activation with light has several key advantages: it can be precisely targeted in time and space, affects only a small area, and doesn’t risk damaging brain tissue.
Photo-activation is made possible by a feat of genetic modification; to make light-sensitive neurons, researchers must genetically engineer them to produce a kind of protein called an “opsin,” the same class of molecules that allow our eyes to see. Genetic instructions for producing opsins can be delivered to neurons via an engineered virus injected into the brain. Once they are installed on the cell membrane, the opsins create a controllable ion channel that excites or inhibits nerve signals when exposed to certain colors of light.
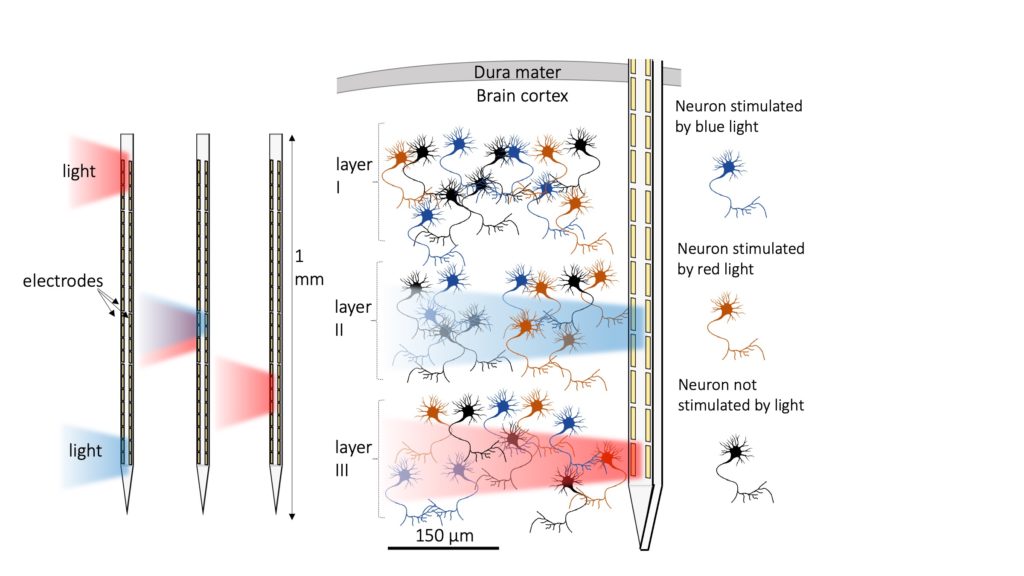
These modified neurons allow extremely fine-scale control over brain activity if light can safely be delivered to the right spot, but historically, that has been a big “if.” Researchers have long had to juggle trade-offs when it comes to probe capabilities, especially when it comes to controlling light without electrical components that produce too much heat. The ideal neural probe would have both multiple light outputs and a large array of detectors, as well as the ability to emit light in different colors and different locations along the probe, all while being small and cool enough to be inserted and operated without causing brain damage. To date, no device has been able to do all of these at once— until now.
Lanzio and the team overcame those hurdles using nano-scale structures called “ring resonators” to control light from a single external laser in a compact, non-electric way. Ring resonators are tiny ring-shaped waveguides, essentially wires that conduct light instead of electricity, that resonate when carrying wavelengths of light that are an exact multiple of their circumference. This property allows them to effectively steal certain wavelengths of light from other waveguides in their vicinity through a process called “coupling.” Once light from an input is captured by a ring resonator, it can be coupled into another waveguide on the opposite side and directed to a reflecting structure that emits it out into the brain.
The new probes take advantage of this redirection by employing a series of ring resonators and emitters along their length like a set of railroad switches. Because ring resonators are only activated by a specific wavelength of light, the operator can choose which position along the probe is being illuminated by making tiny changes to the color of the input laser. In this way, a single input can be split and channeled to different emitters without the use of any bulky or heat-generating components.
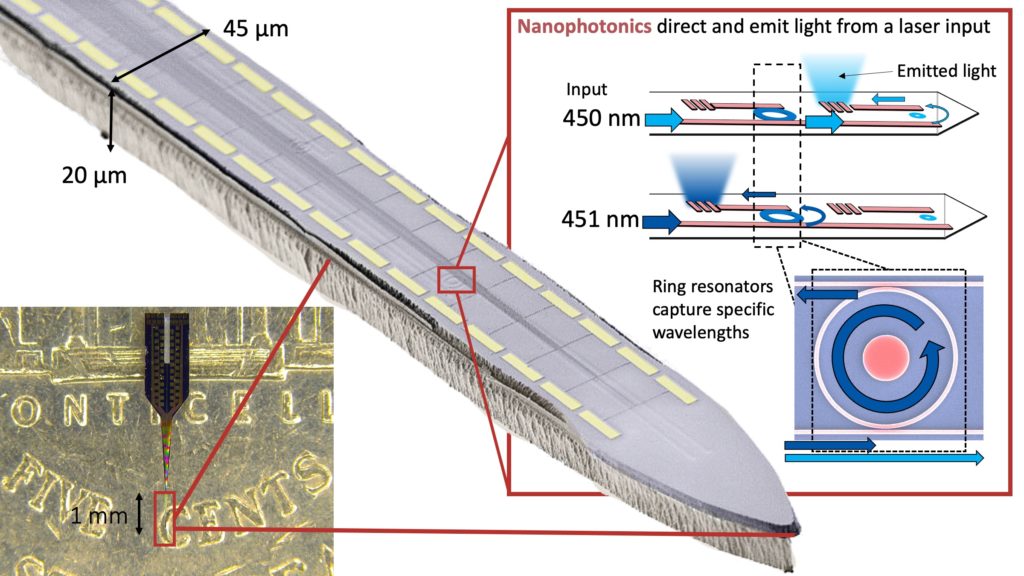
All of this can be accomplished in an exceptionally small device thanks to the advanced nanofabrication capabilities of the Foundry. The waveguides and ring resonators can be etched into a single layer of silicon nitride, and overlain directly with another layer containing the probe’s 64 electrode detectors, a stacked construction that enables these millimeter-long probes to be only 45 millionths of a meter (μm) wide and 25 μm thick.
Interested in Becoming a Foundry User?
Join our collaborative, multidisciplinary environment.
Learn more >
This new level of miniaturization is what most excites the research team. “We now have a way to build much more functionality into these probes without increasing their size,” says Cabrini. “Next we can try to add microfluidics for delivering drugs, or more colors to target different opsins. We proved that 2 layers is possible, let’s try three layers, four layers– let’s see what capabilities can be achieved.”
Even as-is, the probe’s ability to interact with neurons at such a precise scale will enable unprecedented understanding of how signals propagate through the brain’s networks and regions. These advances could pave the way to major benefits for humans, such as new treatments for neurological diseases like Parkinson’s. In the more distant future, the day may even come when this technology is used to interface directly with human brains, allowing real-time insight and control that will bring a powerful new frontier for science and medicine as well as more sinister possibilities for misuse. Lanzio and Cabrini acknowledge that use in humans will require thoughtful oversight and regulations, but also that the medical and technological fields needed to test and implement human applications are still in their infancy. They don’t expect to cross that bridge for some time.
Read the papers:

